Andreas Mund, Fabian Coscia, András Kriston, Réka Hollandi, Ferenc Kovács, Andreas-David Brunner, Ede Migh, Lisa Schweizer, Alberto Santos, Michael Bzorek, Soraya Naimy, Lise Mette Rahbek-Gjerdrum, Beatrice Dyring-Andersen, Jutta Bulkescher, Claudia Lukas, Mark Adam Eckert, Ernst Lengyel, Christian Gnann, Emma Lundberg, Peter Horvath & Matthias Mann
Nature Biotechnology, 2022
Mund et al. have developed a revolutionary method called Deep Visual Proteomics (DVP), which for the first time, allows for the analysis of thousands of proteins from single cells while keeping their original location in the tissue intact. Our Biological Image Analysis Software (BIAS) was a key component of this research, providing the powerful AI-driven image analysis needed to identify and classify cells for proteomic analysis. This groundbreaking work, published in Nature Biotechnology, opens up new avenues for understanding the molecular details of diseases like cancer.
A Deeper Look into Deep Visual Proteomics
For decades, scientists have been able to see cells under a microscope and analyze their proteins separately. However, a major challenge has been to bridge the gap between these two worlds: to know exactly which proteins are in a specific cell that we are looking at. This is particularly important in diseases like cancer, where a tumor is a complex mix of different cell types, each with its own protein signature. The ability to link the visual appearance of a cell with its proteome at a single-cell level can provide unprecedented insights into the disease.
This is where Deep Visual Proteomics (DVP) comes in. The method, developed by Mund et al., is a sophisticated workflow that combines four key technologies:
- High-Resolution Imaging: First, advanced microscopy is used to create detailed maps of the tissue, showing all the different cells in their natural environment.
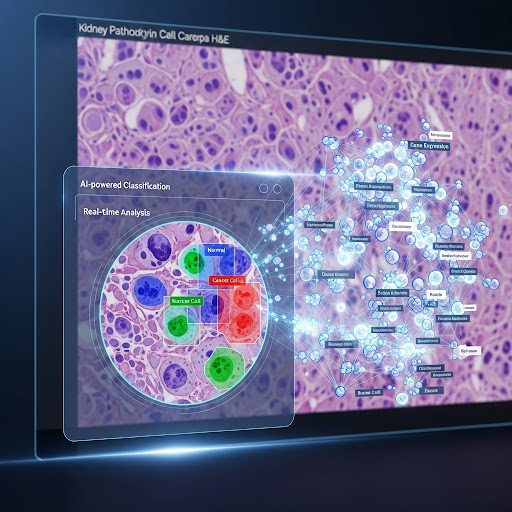
- AI-Powered Image Analysis with BIAS: This is where our software, BIAS, plays a crucial role. BIAS uses artificial intelligence and deep learning to analyze the high-resolution images. It can automatically identify and segment individual cells and even different compartments within the cells, like the nucleus and cytoplasm. Furthermore, BIAS can classify cells into different groups based on their appearance or other features, for example, distinguishing between healthy and cancerous cells.
- Automated Laser Microdissection: Once BIAS has identified the cells of interest, a high-precision laser is used to cut out these single cells from the tissue sample. BIAS serves as the interface here, transferring the exact coordinates of the classified cells to the laser microdissection microscope, ensuring that only the desired cells are collected.
- Ultra-High-Sensitivity Mass Spectrometry: Finally, the collected single cells are analyzed using mass spectrometry, a technique that can identify and quantify thousands of proteins from a tiny amount of material.
By combining these steps, DVP creates a direct link between the visual information from the microscope and the deep molecular data from the proteome.
In their paper, Mund and his team demonstrated the power of DVP by applying it to the study of melanoma. They were able to track the changes in the proteome as normal skin cells (melanocytes) transformed into invasive cancer cells, all within the same piece of tissue. This allowed them to identify specific proteins and cellular pathways that are altered during cancer progression, opening up the possibility of new therapeutic targets.
The development of Deep Visual Proteomics is a significant step forward in the field of spatial biology. It provides a powerful new tool for researchers to explore the complexities of cellular function and disease at an unprecedented level of detail. We at Single-Cell Technologies are proud that our BIAS software was an integral part of this innovative research, and we are excited to see how DVP will be used to make new discoveries in the future.